PLEASE HELP ASAP
Two glasses labeled A and B contain equal amounts of water at different temperatures. Maya put a drop of blue dye into each of the two glasses. The table shows the time taken by the water in the two glasses to become blue.
Dye Experiment
Glass Time
A 5 seconds
B 20 seconds
Which of the following statements is correct?
The water in Glass A is cooler than the water in Glass B; therefore, the particles in Glass A move faster.
The water in Glass A is warmer than the water in Glass B; therefore, the particles in Glass A move faster.
The water in Glass A is cooler than the water in Glass B; therefore, the water particles in Glass A are stationary.
The water in Glass A is warmer than the water in Glass B; therefore, the water particles in Glass A are stationary.
Answers
Answer:
The water in glass A is warmer than the water in glass B therefore the particles move faster
Explanation:
Diffusion is influenced by temperature, concentration gradient and others. in this case the higher the temperature the faster the diffusion rate while when cooler it is slower
Answer:
B. The water in Glass A is warmer than the water in Glass B; therefore, the particles in Glass A move faster.
Explanation:
I took the exam
Related Questions
Which of the following(s) is/are incorrect about the convexity term of a bond:
Group of answer choices
Convexity is always positive for a plain-vanilla bond..
We can improve the estimation of a price change with regard to a change in interest rates by accounting for the convexity of the bond.
Convexity has high value when investors expect that market yields will not change much.
Answers
The correct answer is "Convexity has high value when investors expect that market yields will not change much." This statement is incorrect about the convexity term of a bond.
Convexity is the curvature of the price-yield relationship of a bond and a measure of how bond prices react to interest rate shifts.
Convexity is a term used in bond markets to describe the shape of a bond's yield curve as it changes in response to a shift in interest rates.
Bond traders use the convexity term to estimate the effect of interest rate changes on bond prices more precisely.
Bond traders use the term convexity to measure the rate of change of duration, which is a measure of a bond's interest rate sensitivity.
Convexity term and its features Convexity is always positive for a plain-vanilla bond.
We can improve the estimation of a price change with regard to a change in interest rates by accounting for the convexity of the bond.
Convexity is higher when market yields are unstable or when the bond has more extended maturity and lower coupon rates.
Thus, the correct statement about the convexity term of a bond is:
Convexity is higher when market yields are unstable or when the bond has more extended maturity and lower coupon rates.
To know more about Convexity visit;
https://brainly.com/question/31834216
#SPJ11
Calculate the mass of chromium that can be formed from 1.25 kg of chromium oxide
Answers
The balanced chemical equation for the reaction is:
2 Cr2O3 + 3 Al -> 4 Cr + 3 Al2O3
This equation tells us that two moles of chromium oxide (Cr2O3) react with three moles of aluminum (Al) to produce four moles of chromium (Cr) and three moles of aluminum oxide (Al2O3).
To calculate the mass of chromium that can be formed from 1.25 kg of chromium oxide, we need to use stoichiometry.
First, we need to determine the number of moles of chromium oxide in 1.25 kg of chromium oxide. The molar mass of Cr2O3 is 152 g/mol, so 1.25 kg (or 1250 g) of Cr2O3 is equal to:
1250 g / 152 g/mol = 8.22 mol
According to the balanced chemical equation, 2 moles of Cr2O3 produce 4 moles of Cr. Therefore, 8.22 moles of Cr2O3 will produce:
4/2 x 8.22 mol = 16.44 mol of Cr
Finally, we need to convert the number of moles of chromium produced to mass. The molar mass of Cr is 52 g/mol, so:
16.44 mol x 52 g/mol = 855.36 g
Therefore, the mass of chromium that can be formed from 1.25 kg of chromium oxide is approximately 855.36 grams, or 0.85536 kg.
What is the mass of an object with a density of 2g/mL that displaces 1.5mL of water?
O 0.50
O 1.75
O there is not enough information to tell
O 3
Answers
Answer:
1.75
Explanation:
if you add
Hey besties can y'all help me out
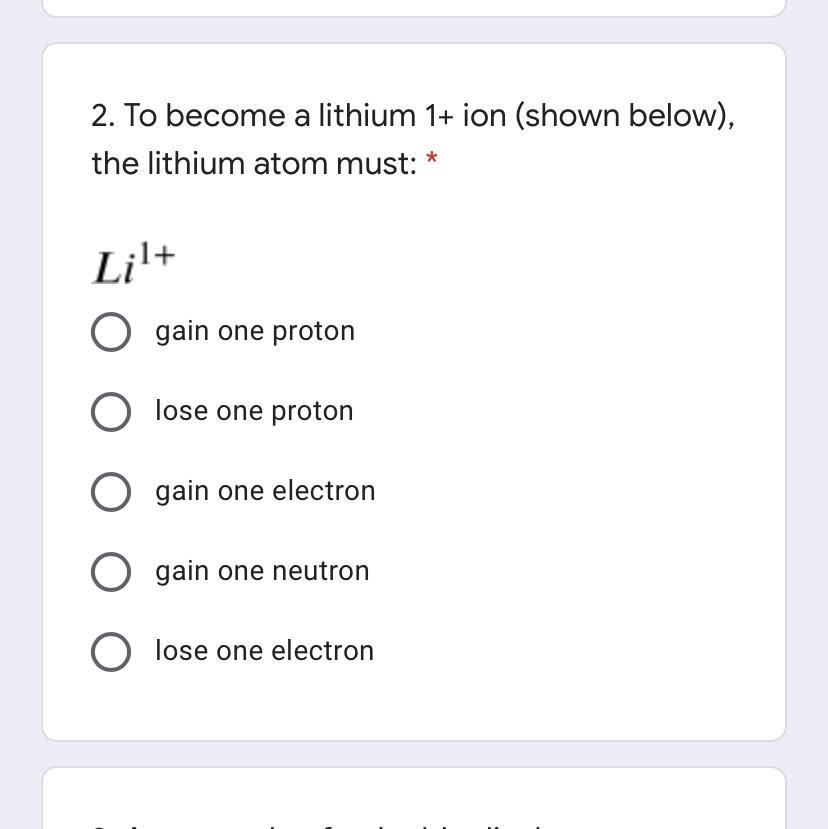
Answers
you need to lose one electron
which would most likely cause a rise in sea levels

Answers
Answer:
B. melting of ice caps
Explanation:
chemists often report percent yield for reactions. what is the formula for calculating percent yield?
Answers
The formula for calculating percent yield is: % Yield = (Actual Yield/Theoretical Yield) x 100
The percent yield of a reaction is a measure of how efficiently a reaction produces its desired product.
It is calculated by dividing the actual yield of the reaction by the theoretical yield of the reaction, and then multiplying by 100 to get the percent yield.
The actual yield is the amount of product that is actually produced by the reaction, while the theoretical yield is the amount of product that would be produced if the reaction were to run perfectly, with no loss of reactants.
For the percent yield, the first step is to measure the actual yield of the reaction. This can be done in the lab by measuring the mass of the product after the reaction is complete.
The actual yield is then divided by the theoretical yield to give the fractional yield of the reaction. The fractional yield is then multiplied by 100 to get the percent yield.
The formula for calculating percent yield is: % Yield = (Actual Yield/Theoretical Yield) x 100. This formula can be used to determine how efficiently a reaction produces its desired product.
to know more about percent yield refer here:
https://brainly.com/question/17042787#
#SPJ11
Answer: (Actual Yield/Percent Yield)*100
Explanation:
The actual yield is the amount of product formed in a reaction.Theoretical yield is the maximum amount of possible product formed in a reaction if the limiting reactant is completely converted to product. Percent yield is calculated as (moles of actual yield divided by moles of theoretical yield)*100.
freshly mixed concrete is not a liquid but a slurry, a semistable mixture of solids suspended in liquid. care must be taken to prevent the material from segregating, creating nonuniform and generally unsatisfactory properties. which of the following will not cause segregation to occur?
Answers
Following things will not cause segregation to occur - By adding water slowly and evenly, controlling the slump of the concrete mixture, and limiting the travel distance from the mixing plant to the job site.
Freshly mixed concrete is not a liquid but a slurry, a semistable mixture of solids suspended in liquid. Care must be taken to prevent the material from segregating, creating nonuniform and generally unsatisfactory properties.
Adding water slowly and evenly ensures that the water does not seep away to one end of the concrete, resulting in the concrete drying in patches. A high water to cement ratio increases the slump of the concrete mixture, making it flow freely.
This makes it easier for the water to spread out evenly throughout the mix, eliminating the risk of segregation. Limiting the travel distance from the mixing plant to the job site reduces the time the concrete is exposed to wind, rain, and other external factors which can cause the water to settle unevenly, resulting in segregation.
Know more about segregation here:
https://brainly.com/question/19904337
#SPJ11
Please help me answer this it’s due today. I will give brainliest

Answers
Answer: X should represent H, hydrogen.
Explanation:
The H is the only one that hasnt been stated in the left side of the formula. H has three atoms as well.
predict whether the h2so4, hso4- is conjugate acid/base pair or not. group of answer choices yes no
Answers
The H₂SO₄ (sulfuric acid) and HSO₄⁻ (hydrogen sulfate or bisulfate ion) are considered a conjugate acid-base pair. The correct answer is yes.
H₂SO₄ (sulfuric acid) and HSO₄⁻ (hydrogen sulfate or bisulfate ion) form a conjugate acid-base pair. In the context of the Bronsted-Lowry theory, an acid donates a proton (H+), while a base accepts a proton. When H2SO4 donates a proton, it becomes HSO₄⁻.
Conversely, when HSO₄⁻ accepts a proton, it reforms H₂SO₄. They are interconnected through the transfer of a proton, thus qualifying as a conjugate acid-base pair. This relationship allows for the reversible conversion between the two species through proton transfer reactions. Therefore, yes, H₂SO₄ and HSO₄⁻ are considered a conjugate acid-base pair.
To learn more about Bronsted-Lowry theory visit: https://brainly.com/question/15516010
#SPJ11
You're paid $25 per hour for your job. How much would you earn in cents per second?
Answers
Answer:
0.694 cents per second
Explanation:
25x100=2500 cents per hour, 2500/60 = 41.67 per minute and 41.67/60=0.694 cents per second
Which substance is composed of only one type of atom? - Need asap!
Water
Gold
Salt
Sugar
Answers
Answer:
Gold
Hope it helps
have a good day
What is the name of the compound - Ca3As2?
Answers
Answer:
Calcium Arsenide
Aspirin (C9H8O4) is an acid which can be titrated with a base to determine purity. If an aspirin tablet weighing 0.615 g is titrated with standardized 0.1121 M KOH, the endpoint is reached after 20.52 mL of KOH have been added. What is the percentage of aspirin in the tablet
Answers
Answer:
67.4 % of C₉H₈O₄
Explanation:
To make titrations problems we know, that in the endpoint:
mmoles of acid = mmoles of base
mmoles = M . volume so:
mmoles of acid = 20.52 mL . 0.1121 M
mmoles of acid = mg of acid / PM (mg /mmoles)
Let's determine the PM of aspirin:
12.017 g/m . 9 + 1.00078 g/m . 8 + 15.9994 g/m . 4 = 180.1568 mg/mmol
mass (mg) = (20.52 mL . 0.1121 M) . 180.1568 mg/mmol
mass (mg) = 414.4 mg
We convert the mass to g → 414.4 mg . 1g / 1000mg = 0.4144 g
We determine the % → (0.4144 g / 0.615 g) . 100 = 67.4 %
eleanor is randomly choosing a pair of socks from her dresser. she has 6 pairs of white socks, 8 pairs of black socks, and 14 pairs of gray socks. complete the choice matrix by determining whether each statement is true or false.
Answers
The choice matrix for Eleanor's sock selection, considering the terms "randomly choosing" and "choice matrix".
1. True
2. False
3. True
4. True
Statement 1: There is an equal chance of choosing any pair of socks.
True. Since Eleanor is randomly choosing a pair of socks, each pair has an equal chance of being chosen, regardless of its color.
Statement 2: The probability of choosing white socks is greater than choosing black socks.
False. Eleanor has 6 pairs of white socks and 8 pairs of black socks. Since there are fewer white socks, the probability of choosing white socks is lower than choosing black socks.
Statement 3: The probability of choosing gray socks is the highest.
True. Eleanor has 14 pairs of gray socks, which is more than the other colors. Therefore, the probability of choosing gray socks is the highest.
Statement 4: The sum of the probabilities of choosing white, black, and gray socks is 1.
True. Since the probabilities of all possible outcomes should add up to 1, the sum of the probabilities of choosing white, black, and gray socks is 1.
For more question on choice matrix : https://brainly.com/question/20847653
#SPJ11
the third law of thermodynamics describes the entropy of a: select the correct answer below: solid liquid gas all of the above
Answers
The third law of thermodynamics describes the entropy of a: solid.
The third law of thermodynamics states that the entropy of a pure crystalline substance approaches zero as the temperature approaches absolute zero (0 Kelvin or -273.15 degrees Celsius). This law implies that at absolute zero, a perfectly ordered and pure crystalline solid will have zero entropy.
The third law of thermodynamics is not specific to liquids or gases but applies to solids. In a solid, the molecules are highly ordered and have fixed positions in a regular lattice structure. As the temperature decreases towards absolute zero, the thermal motion of the molecules reduces, and the system becomes more ordered, resulting in a decrease in entropy.
In contrast, liquids and gases have higher entropy compared to solids at absolute zero because their molecules have more freedom of movement and are not as tightly arranged. Therefore, the third law of thermodynamics specifically addresses the entropy of solids and does not apply to liquids or gases.
To learn more about law of thermodynamics, here
https://brainly.com/question/1368306
#SPJ4
Place these unknown pH test papers in order from most acidic to most alkaline.
Four pH indicator strips.
Answers
Answer:
In order of the colors of the pH test papers from most acidic solutions to most alkaline solutions, the colors of the pH test papers would be
Red > Orange/Pink > Purple > Blue
(Red) Most acidic (Blue)Most alkaline
Explanation:
We can use the different colors of pH test papers to determine the acidity or alkalinity of a solution.
Definitions
A pH test paper is a strip of indicator paper which changes colour when placed in acidic or alkaline solutions.
A solution is said to be acidic if it turns the colour of the pH test paper RED
A solution is said to be alkaline/basic if it turns the colour of the pH test paper BLUE
Color changes
If the pH test paper turns red, then the solution is a very strongly acidic solution with pH less than 3 ( ie pH between 1-3)
If the pH test paper turns orange or pink, then the solution is a weak acidic solution with pH greater than 3 ut less than 7
If the pH test paper turns purple, then the solution is a weak alkaline solution with pH greater between 8-11
If the pH test paper turns blue, then the solution is a very strong alkaline solution with pH between 11 - 14
Summary
Therefore, in order of color changes from the most acidic solutions to most alkaline solutions, the colors of the pH test papers would be
Red > Orange/Pink > Purple > Blue
Most acidic Most alkaline
Answer:
d<c<a<b
Explanation:
which compound below will readily react with a solution of bromine consisting of 48% hydrobromic acid and 30% hydrogen peroxide? group of answer choicesacetic acidcyclohexanedichlorometanet-butyl alcoholcyclohexene
Answers
The compound that will readily react with a solution of bromine consisting of 48% hydrobromic acid and 30% hydrogen peroxide is acetic acid.Hydrobromic acid is a solution of hydrogen bromide (HBr) in water.
It is a strong acid that can be used for several industrial purposes. Hydrogen peroxide is a reactive chemical with the chemical formula H2O2. When combined with hydrobromic acid, hydrogen peroxide forms a solution known as hydrobromic acid. Bromine is a non-metallic element that reacts with many compounds to form new substances.Acetic acid (CH3COOH) is a weak organic acid that is commonly found in vinegar and is used in the production of cellulose acetate.
When acetic acid is mixed with a solution of bromine containing 48% hydrobromic acid and 30% hydrogen peroxide, it reacts readily. This reaction will produce a new compound.The other three options (cyclohexane, dichloromethane, t-butyl alcohol, and cyclohexene) do not have any active functional groups such as an alcohol, carboxylic acid, or an unsaturated bond that can undergo halogenation or oxidation reactions. Therefore, they will not react with a solution of bromine consisting of 48% hydrobromic acid and 30% hydrogen peroxide.
For more such questions on Acetic acid
https://brainly.com/question/24304533
#SPJ8
Provide any critical observations/changes to the reaction mixture upon addition of the acid. Propose what this might suggest about possible functional groups present on your unknown starting material and briefly defend your proposal. A white precipitate forms with each drop of sulfuric acid.
Answers
This suggests that the unknown starting material is likely an organic compound containing a carboxylic acid functional group.
What is compound ?Compound is a combination of two or more elements which are chemically bonded together. Compounds can exist in both organic and inorganic forms, and are formed when atoms of different elements combine to form molecules. Inorganic compounds are generally composed of metal and non-metal elements, while organic compounds are composed of only carbon and hydrogen atoms. Compounds can also be classified according to their physical state, such as solid, liquid, or gas. Compounds are important in many areas of science, such as chemistry, physics, and biology, and play an essential role in the production of many products, including medicines, plastics, and fuels.
To learn more about compound
https://brainly.com/question/29108029
#SPJ4
give the systematic name for the compound ba no3 2
Answers
The systematic name for the compound Ba(NO3)2 is barium nitrate. Barium nitrate is an inorganic salt with the chemical formula Ba (NO3)2. It is a colorless, odorless, and crystalline solid that is highly soluble in water. The compound is formed by combining one atom of barium and two ions of nitrate.
The name “barium” comes from the Greek word “barys,” which means “heavy,” and is a reference to its high density. The term “nitrate” refers to the polyatomic ion NO3-, which is composed of one nitrogen atom and three oxygen atoms. Barium nitrate is commonly used in pyrotechnics, as it is a powerful oxidizing agent that produces a bright green flame when ignited.
The systematic naming of inorganic compounds is based on the rules set out by the International Union of Pure and Applied Chemistry (IUPAC). The name of an ionic compound is composed of the cation name followed by the anion name. In the case of barium nitrate, “barium” is the name of the cation, while “nitrate” is the name of the anion.
Therefore, the systematic name for the compound Ba(NO3)2 is barium nitrate.
Know more about systematic name here:
https://brainly.com/question/1787202
#SPJ11
solar and wind power are ____ and safe forms that are renewable and don't pollute
Answers
Which terin refers to the attraction of water molecules to one another? a. adhesion b. cohesion c. hydrolysis d. photolysis e. polarity
Answers
The term that refers to the attraction of water molecules to one another is "cohesion". The correct answer is option: b. cohesion.
Cohesion is a property of water molecules that arises from their polarity and hydrogen bonding. The oxygen atom in each water molecule is partially negative, while the hydrogen atoms are partially positive, creating a polar molecule. The polar nature of water allows the oxygen atoms in one molecule to form hydrogen bonds with the hydrogen atoms in nearby water molecules, resulting in a strong attraction between them. This cohesive property of water is responsible for many of its physical properties, such as surface tension and capillary action. Option b is correct.
To know more about Cohesion, here
brainly.com/question/29598400
#SPJ4
The system: N2O (g) + NO2(g)
⇔
3 NO (g) is at equilibrium. You add some NO2 and allow the system to adjust to the change. For each substance, indicate whether the amount of that substance present has increased, decreased or not changed when compared to the amount present in the original equilibrium.

Answers
When \(NO2\) is added to the system, according to Le Chatelier's principle, the equilibrium will shift to counteract the increase in\(NO2\). The reaction will proceed in the forward direction to consume the excess \(NO2\).
As a result, the amount of \(N2O\) will decrease, the amount of \(NO2\) will decrease, and the amount of\(NO\)will increase. The equilibrium will shift to the right to maintain a constant value of Kc.
Therefore, the amount of \(N2O\) and \(NO2\)will decrease, and the amount of \(NO\) will increase. This is because the forward reaction \((N2O + NO2 = 3NO)\)will consume the added\(NO2\), which will cause the amount of\(N2O\)to decrease. In response, the reverse reaction \((3NO → N2O + NO2)\)will proceed, causing the amount of \(NO\) to increase. The equilibrium will shift in the forward direction to restore the balance between the reactants and products.
Learn more about equilibrium here
https://brainly.com/question/30807709
#SPJ1
how many grams are in 2.34 moles of Li?
Answers
Answer:
The SI base unit for amount of substance is the mole. 1 mole is equal to 1 moles Lithium, or 6.941 grams.
Thus, the answer would be 16.24194.
Explanation:
please help me. :)))

Answers
46.12 grams of water are produced when 35 grams of \(C_6H_1_0\) react with 45 grams of \(O_2\).
Stoichiometric problemThe balanced equation for the combustion of C6H10 (cyclohexene) with O2 is:
\(C_6H_{10} + O_2 - > CO_2 + H_2O\)
From the balanced equation, we can see that 1 mole of \(C_6H_1_0\) reacts with 6 moles of O2 to produce 6 moles of water.
First, let's calculate the number of moles of C6H10 and O2:
Molar mass of C6H10 = 6(12.01 g/mol) + 10(1.01 g/mol) = 82.16 g/mol
Number of moles of C6H10 = mass / molar mass = 35 g / 82.16 g/mol ≈ 0.426 mol
Molar mass of O2 = 2(16.00 g/mol) = 32.00 g/mol
Number of moles of O2 = mass / molar mass = 45 g / 32.00 g/mol ≈ 1.406 mol
From the stoichiometry of the balanced equation, we can determine that 0.426 moles of C6H10 will produce 0.426 * 6 = 2.556 moles of water.
Now, let's calculate the mass of water produced:
Molar mass of H2O = 2(1.01 g/mol) + 16.00 g/mol = 18.02 g/mol
Mass of water = number of moles of water * molar mass of water
= 2.556 mol * 18.02 g/mol
≈ 46.12 g
Therefore, approximately 46.12 grams of water are produced when 35 grams of C6H10 reacts with 45 grams of O2.
More on stoichiometric problems can be found here: https://brainly.com/question/32305503
#SPJ1
I need the answer asap

Answers
Answer:
The energy will be added
Explanation:
When the body becomes overheated, how does the body react to maintain homeostasis?
The nervous system directs your body to sweat to release heat.
The digestive system slows down to save energy and release heat.
d
The respiratory system directs your lungs to breath more to increase blood flow and release heat.
The muscular system signals your body to shake and shiver and hair follicles stand on end to release heat.
Answers
Answer:
Nervous system directs your body to swear to release heat.
Explanation:
Sweat maintains homeostasis when overheating.
When the body becomes overheated, the nervous system directs your body to sweat to release heat. Cooling effect of sweating over body balance the homeostasis.
What is homeostasis ?Any self-regulating mechanism called homeostasis helps biological systems to maintain stability while responding to environmental factors that are appropriate for survival. If homeostasis is attained, life goes on; if it is not, tragedy or death result.
The stability obtained is actually a dynamic equilibrium, where circumstances are mostly uniform yet change is constant. The ability to regulate their body temperature is a prime example of homeostasis in a biological system.
Humans typically have body temperatures that vary about 37 °C, but a number of variables, such as exposure, hormones, metabolic rate, and sickness, can alter this value and cause abnormally high or low temperatures. The sweating is a good example of homeostasis.
Find more on homeostasis :
https://brainly.com/question/2826402
#SPJ2
Help me please !!!
Which of the following can be inferred from the diagram above that shows the dependence of potential Energy on the Internuclear distance between two atoms?
A)The atoms form a bond with a bond length of 25 pm
B)The atoms form a bond with a bond length of 75 pm
с)The net force between the atoms is attractive at 25 pm
D)The net force between the atoms is attractive at 75 pm

Answers
Answer:
Its B. The atoms form a bond with a bond lenth of 75 pm
If P increases by a factor of 5 and T decreases by a factor of 3, what will be the change in V?
A. V increases by a factor 3
B. V increases by a factor of 15
C. V decreases by 3/5
D. V increases by a factor 5
E. V decreases by a factor of 15
Answers
\( \dag \: \: \: \huge{ \boxed{ \sf{ \pink{A\green{N \blue{S\color{yellow}W\red{E\orange{R}}}}}}}}\)
To determine the change in volume (V) given the changes in pressure (P) and temperature (T), we need to consider the relationship described by the ideal gas law:
\(\displaystyle PV = nRT\)
Where:
- P is the pressure- V is the volume- n is the number of moles of gas- R is the ideal gas constant- T is the temperatureTo analyze the effect of changing P and T on V, we'll assume that the number of moles (n) and the ideal gas constant (R) remain constant.
If P increases by a factor of 5, we can express the new pressure as:
\(\displaystyle P_{\text{new}} = 5P\)
If T decreases by a factor of 3, we can express the new temperature as:
\(\displaystyle T_{\text{new}} = \frac{1}{3}T\)
Now, let's consider the relationship between the initial and final volumes (V and V_new):
\(\displaystyle PV = nRT\)
\(\displaystyle V = \frac{nRT}{P}\)
\(\displaystyle P_{\text{new}}V_{\text{new}} = nRT_{\text{new}}\)
\(\displaystyle V_{\text{new}} = \frac{nRT_{\text{new}}}{P_{\text{new}}}\)
Substituting the expressions for P_new and T_new, we have:
\(\displaystyle V_{\text{new}} = \frac{nR\left(\frac{1}{3}T\right)}{5P}\)
Simplifying the expression:
\(\displaystyle V_{\text{new}} = \frac{1}{15}\left(\frac{nRT}{P}\right)\)
Comparing this with the initial volume (V), we can see that:
\(\displaystyle V_{\text{new}} = \frac{1}{15}V\)
Therefore, the change in volume (V) is such that it decreases by a factor of 15.
The correct option is E. V decreases by a factor of 15.
\(\huge{\mathfrak{\colorbox{black}{\textcolor{lime}{I\:hope\:this\:helps\:!\:\:}}}}\)
♥️ \(\large{\underline{\textcolor{red}{\mathcal{SUMIT\:\:ROY\:\:(:\:\:}}}}\)
commonly held belief is that you can boil water faster by adding salt (a nonvolatile solute). in light of what we have just learned would you predict this belief to be true? if not, what impact on the rate of boiling water would you predict? prof. josh colligative properties 25 / 30
Answers
The commonly held belief that you can boil water faster by adding salt (a nonvolatile solute) is not true.
According to the concepts of colligative properties, the boiling point of a solution is increased by the addition of a nonvolatile solute. This implies that the boiling point of saltwater should be higher than that of pure water, not lower.
However, salt does have an impact on the rate of boiling water. The addition of salt to water, particularly a small quantity, has no effect on the boiling process or on the temperature at which it occurs. The reason for this is that adding salt to water does not reduce the amount of heat required to boil it, so it boils at the same temperature as pure water
.However, when salt is added to water in greater amounts, the boiling temperature of the water increases. In other words, the more salt that is added to water, the higher the boiling temperature becomes. This is due to the fact that adding a nonvolatile solute raises the boiling point of the solution.
Learn more about solution at:
https://brainly.com/question/29454798
#SPJ11
when precipitating the lead cation, you were told to place the test tube in ice after adding the appropriate reagent. suggest a possible reason for this instruction.
Answers
When precipitating the lead cation, you were told to place the test tube in ice after adding the appropriate reagent because this helps to increase the solubility of the lead cation and allows for more efficient precipitation.
By lowering the temperature of the solution, the solubility of the lead cation is decreased, causing it to precipitate out of solution more readily. This is known as the "common ion effect" and is a common technique used in chemistry to promote precipitation reactions.
By placing the test tube in ice, the reaction is slowed down, allowing for more complete precipitation of the lead cation and resulting in a more accurate measurement of the amount of lead present in the solution.
To know more about solubility click here:
https://brainly.com/question/29661360#
#SPJ11